
*low boiling points Allotropes two or more different molecular forms of the same element in the same physical state ionization energy The amount of energy required to remove an electron from an atom atomic radius one-half the distance between the nuclei of two atoms of the same element when the atoms are joined ionic radius Distance from the center of an ion's nucleus to its outermost electron Electronegativity A measure of the ability of an atom in a chemical compound to attract electrons Reactivity How readily a substance combines chemically with other substances. 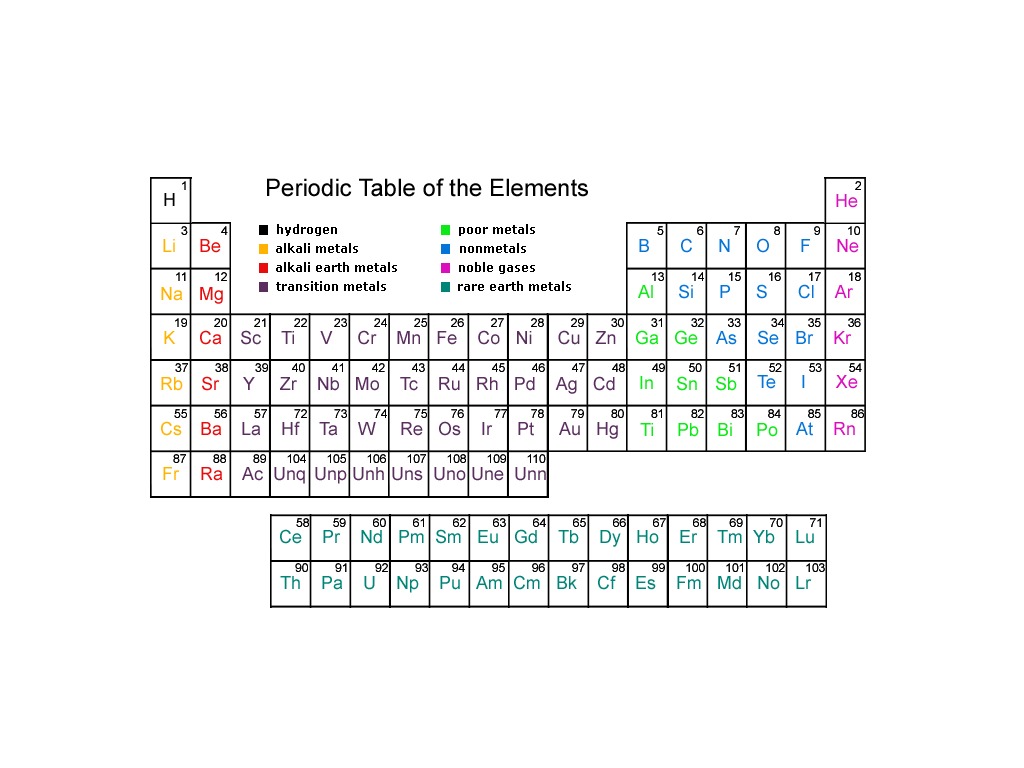
Non-metals Low conductivity, not ductile, not malleable, brittle, dull, gas at room temp noble gases Group 18: You will be provided with a periodic table and a. Along the staircase between metals and nonmetals and have both metallic and nonmetallic properties. Topics covered in this course include Atomic Structure, Periodic Table, Moles and Stoichiometry, Chemical Bonding, Physical Behavior of Matter, Kinetics and. AP Chemistry Course and Exam Description This is the core document for the course. Reviews the top 200 concepts in chemistry. Finish all 50 in the set to study for your chemistry regents. transition metals Groups 3-12, 1-2 electrons in the outer energy level, less reactive than alsali-earth metals, shiny, good conductor of thermal energy and electrical current, high density Metalloids B, Si, Ge, As, Sb, Te. These are actual chemistry regents questions from the dated exam digitized and turned into review games for a fun way to review for your regents. Metals Most elements of the periodic table they are typically solid, shiny, malleable, and good conductors of heat and electricity.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
